UVA Author: Douglas DeSimone
Citation: Watanabe H, Belyea BC, Paxton RL, et al. Renin Cell Baroreceptor, a Nuclear Mechanotransducer Central for Homeostasis. Circ Res. 2021;129(2):262-276. doi:10.1161/CIRCRESAHA.120.318711
DOI: https://www.ahajournals.org/doi/10.1161/CIRCRESAHA.120.318711
Pub-Med Number: 33993729
Rationale:
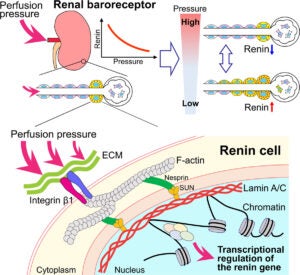
Renin-expressing cells are myoendocrine cells crucial for survival. They have been postulated to possess a pressure-sensing mechanism, a baroreceptor, which can detect slight changes in blood pressure and respond with precise and synchronized amounts of renin synthesized and released to the circulation to maintain blood pressure and fluid-electrolyte balance. The location and nature of this puzzling pressure-sensing structure have remained unknown as it was originally suggested over 60 years ago.
Objective:
To elucidate the location and structure of the renin cell baroreceptor.
Methods and Results:
We used a variety of genetically modified mice whereby renin cells were exposed in vivo to either low or high arterial pressure. In addition, we applied direct mechanical stimuli, that is, pneumatic pressure or stretch, directly to renin cells cultured under different conditions and substrata. Changes in perfusion pressure and/or direct mechanical stimuli induced significant changes in renin gene expression and the phenotype of renin cells. Importantly, the experiments show that the pressure-sensing mechanism (the baroreceptor) resides in the renin cells; it requires initial extracellular sensing by integrin β1 at the renin cell membrane and is transduced to the nuclear membrane and chromatin by lamin A/C.
Conclusions:
These studies show that the enigmatic baroreceptor is a nuclear mechanotransducer that resides in the renin cells per se and is responsible for the sensing and transmission of extracellular physical forces directly to the chromatin of renin cells via lamin A/C to regulate renin gene expression, renin bioavailability, and homeostasis.
