Sanja Arandjelovic, PhD
PRIMARY APPOINTMENT

Assistant Professor, Nephrology
Contact:
Center for Immunity, Inflammation, and Regenerative Medicine
UVA Division of Nephrology
PO Box 800133
Charlottesville, Virginia 22908
Email: sa2h@virginia.edu
EDUCATION AND TRAINING
- BS, University of Belgrade (Molecular Biology and Physiology), Belgrade, Serbia
- PhD, University of Virginia (Biochemistry and Molecular Genetics) Charlottesville, VA
- Postdoctoral Fellow (Immunology), The Scripps Research Institute, La Jolla, CA
- Research Scientist (Immunology), University of Virginia
RESEARCH AREAS
- Innate immunity, Cell clearance, Inflammatory and autoimmune disease
RESEARCH SUMMARY
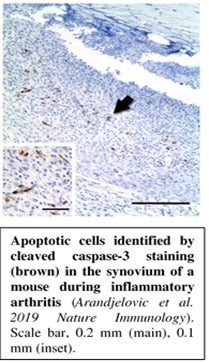
The Arandjelovic laboratory focuses on cell clearance in inflammatory disease, using rheumatoid arthritis (RA) and multiple sclerosis (MS) as model systems. Inflammatory states are associated with high rates of cell death, mostly through apoptosis, and the clearance of apoptotic cells is a critical step in disease pathology. However, the cellular identity of phagocytes and the molecular events that contribute to the clearance of apoptotic cells has been understudied in many inflammatory diseases. In other words, “who” are the cleaning crew and which “tools” are they using?
Current projects:
The function of the engulfment regulator ELMO1 in inflammation. Cell clearance regulator engulfment and cell motility-1 (ELMO1) contribute to neutrophil function in arthritis but are dispensable in several other inflammatory states, including lung and peritoneal inflammation. In addition, the human ELMO1 single nucleotide polymorphism (SNP) rs11984075, which occurs in up to 25% of the human population, is associated with increased neutrophil migratory capacity and higher susceptibility to RA. In this project, we aim to address the tissue specificity of ELMO1 function in neutrophils and identify the consequence(s) of the arthritis-associated ELMO1 rs11984075 on neutrophil functions.
Disease contribution and therapeutic potential of cell clearance pathways in autoimmune inflammation. Apoptotic cells are frequently observed at sites of ongoing inflammation, and inefficient clearance of apoptotic cells can lead to secondary necrosis and the release of their cellular contents, which can further fuel the inflammatory process. However, the nature of dying cells and phagocytes that clear them remains unclear. The questions that we aim to address in this project include:
- Who are the phagocytes in inflamed tissues in RA and MS?
- What are they “eating” (infiltrating leukocytes, dying tissue-resident cells)?
- How does the modulation of engulfment pathways impact disease parameters?
Our studies seek to identify new modulators of inflammatory and autoimmune disease and uncover novel pathways that can be targeted for therapeutic benefit.







