Assembly of functional caveolae
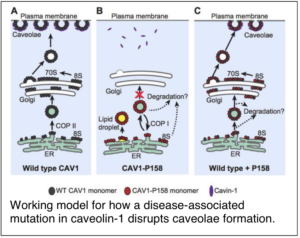
Flask-shaped invaginations of the plasma membrane known as caveolae were first discovered in the 1950s. Since then, caveolae and their major structural protein caveolin-1 have been implicated in numerous cellular events including signaling, mechanosensing, and membrane buffering. Yet extensive study, it remains unclear how caveolae mediate these processes. We helped to clarify potential mechanisms by which caveolin-1 and caveolae control cellular functions by showing that caveolin-1 is particularly sensitive to overexpression and tagging. We also characterized two disease-associated mutants of caveolin-1 that contribute to the fatal lung disease pulmonary arterial hypertension. These mutant forms of caveolin are capable of forming caveolae together with wild type caveolin-1, raising interesting questions about what defines a functional caveola.
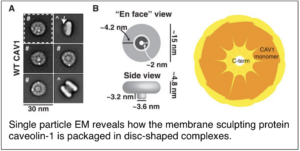
Our most recent work has now set out to answer one of the most long-standing questions in the field: what is the structural basis for caveolae formation and function? To address this question, we have teamed up with the Ohi lab at the University of Michigan to visualize the packaging of caveolin-1 within disc-shaped complexes that serve as a fundamental building block of caveolae using single particle electron microscopy (EM). Efforts to extend this work to obtain a high resolution cryoEM structure of caveolin-1 complexes are currently underway.
Recent publications
Han, B.#, J. C. Porta#, J. L. Hanks, Y. Peskova, E. Binshtein, K. Dryden, D. P. Claxton, H. S. Mchaourab, E. Karakas, M. D. Ohi*and A. K. Kenworthy* (2020) Structure and assembly of caveolin-1 8S complexes revealed by single particle electron microscopy. Sci Adv 6: eabc6185. PMCID: PMC7821874. #equal contributions; *co-corresponding authors.
Rathinasabapathy, A., C. Copeland, A. Crabtree, E. J. Carrier, C. Moore, S. Shay, S. Gladson, E. D. Austin, A. K. Kenworthy, J. E. Loyd, A. R. Hemnes and J. D. West (2020) Expression of a Human Caveolin-1 Mutation in Mice Drives Inflammatory and Metabolic Defect-Associated Pulmonary Arterial Hypertension. Front Med (Lausanne) 7: 540. PMCID: PMC7516012
Parton, R. G., M. A. Del Pozo, S. Vassilopoulos, I. R. Nabi, S. Le Lay, R. Lundmark, A. K. Kenworthy, A. Camus, C. M. Blouin, W. C. Sessa and C. Lamaze (2020) Caveolae: The FAQs. Traffic 21(1): 181-185. PMCID: PMC7073450
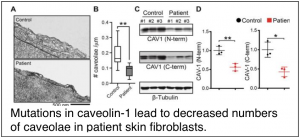
Copeland, C. A. *, B. Han*, A. Tiwari, E. D. Austin, J. E. Loyd, J. D. West, and A. K. Kenworthy (2017) A disease-associated frameshift mutation in caveolin-1 disrupts caveolae formation and function through introduction of a de novo ER retention signal. Mol. Biol. Cell. 28: 3095-3111. PMCID: PMC5662265 *equal contributions
Han, B.*, C. A. Copeland*, Y. Kawano, E. B. Rosenzweig, E. D. Austin, L. Shahmirzadi, S. Tang, K. Raghunathan, W. K. Chung, and A. K. Kenworthy (2016) Characterization of a caveolin-1 mutation associated with both PAH and congenital generalized lipodystrophy. Traffic 17 (12): 1297-1312; doi: 10.1111/tra.12452. PMCID: PMC5197452 *equal contributions
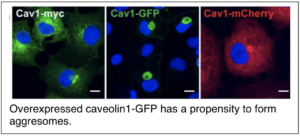
Tiwari, A., C.A. Hanson, B. Han, C.A. Copeland, K. Raghunathan, and A. K. Kenworthy (2016) Caveolin-1 is an aggresome-inducing protein. Scientific Reports 6: 38681. PMCID: PMC5144149
Han, B., C. A. Copeland, A. Tiwari, and A. K. Kenworthy (2016) Assembly and turnover of caveolae: what do we really know? Front Cell Dev Biol, Jun 27;4:68. doi: 10.3389/fcell.2016.00068. eCollection 2016. PMCID: PMC4921483
Prewitt, A.R., S. Ghose, A.L. Frump, A. Datta, E.D. Austin, A.K. Kenworthy, and M.P. de Caestecker (2015) Heterozygous Null Bone Morphogenetic Protein Receptor Type 2 Mutations Promote SRC Kinase-dependent Caveolar Trafficking Defects and Endothelial Dysfunction in Pulmonary Arterial Hypertension. J Biol Chem. 290: 960-71. PMCID: PMC4294523
Han, B., A. Tiwari and A. K. Kenworthy (2015) Tagging strategies strongly affect the fate of overexpressed caveolin-1. Traffic 16(4): 417-38. PMCID: PMC4440517
Hanson, C. A., K. R. Drake, M. A. Baird, B. Han, L. J. Kraft, M. W. Davidson and A. K. Kenworthy (2013) Overexpression of caveolin-1 is sufficient to phenocopy the behavior of a disease-associated mutant. Traffic 14(6): 663-77. PMCID: PMC3674505
