Sho Morioka, PhD
PRIMARY APPOINTMENT

Assistant Professor of Medicine, Nephrology
Contact:
UVA Division of Nephrology & CIIR
PO Box 800133
Charlottesville, VA 22908
Telephone: 919-917-8245
Email: sm9ss@virginia.edu
Pub Med
NIH RePORTER
RESEARCH AREAS
Role of programmed cell death in kidney injury
RESEARCH SUMMARY
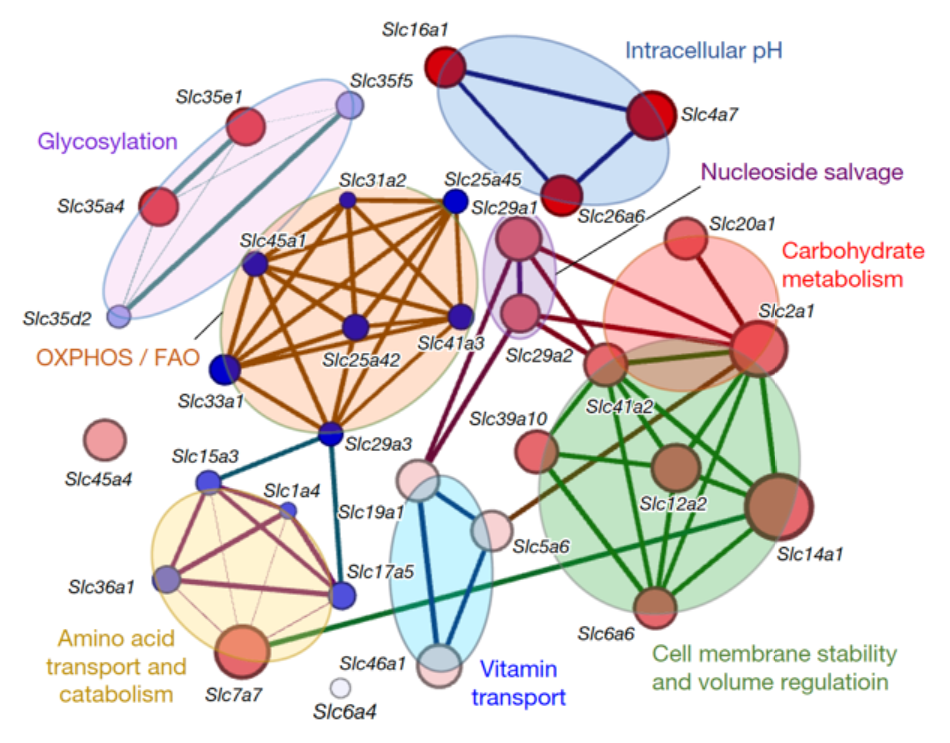
My primary research focus includes regulating programmed cell death (e.g., necroptosis, pyroptosis, and apoptosis). After a brief post-doctoral stint in my graduate thesis lab to complete some projects and publications (Blood 2012, Journal of Cell Biology 2014, Oncogene 2016), I moved to the University of Virginia as a Senior Research Scientist, focusing on identifying the endogenous regulatory pathways that control clearance of apoptotic cells (also termed ‘efferocytosis’). Removal of apoptotic cells by phagocytes occurs at a rate of nearly one million cells per second in the body, and defective clearance leads to inflammatory diseases; yet, there are still significant knowledge gaps in how a phagocyte achieves rapid corpse uptake and how the phagocytosis handles this stress on various aspects of its physiology such as energy metabolism, pH regulation, or volume regulation. I discovered that a novel gene program, the solute carrier (SLC) program, is essential to successfully uptake apoptotic cells (Nature 2018, Immunity 2019, and Nature Cell Biology 2019). During acute kidney injuries (AKI), it is known that phagocyte actively removes apoptotic cells. My current research focuses on the effect of enhancing efferocytosis on the resolution of AKI. I am elucidating molecular mechanisms that enable phagocytes to efficiently remove apoptotic cells under kidney injury using a uniquely created super-engulfed transgenic mouse model.
| Funded Research | Dates |
|---|---|
| NIH/NIDDK 1K01DK123497-01 - Rremoval of Apoptotic Cells During Acute Kidney Injury | 3/20/20-2/28/25 |
| ASN Carl W. Gottschalk Grant. Boosting Removal of Apoptotic Cells during Acute Kidney Injury | 7/1/2020-6/30/2022 |
| UAB-UCSD O’Brien Center Grant- Boosting Removal of Apoptotic Cells During Acute Kidney Injury | 8/1/2020-7/31/2021 |
| Yamada Bee Company Co Inc., Research Contract | 9/1/2019- |


