Other Modification – CTA Amendments
Other Modification - CTA Amendments
| Field | Instructions | Example | Tips/Notes | Screenshot |
|---|---|---|---|---|
| Short Title | Create a short description of the request that is identifiable in the Award Workspace. Do not use "MD-SURG-Smith" naming convention. | "Budget Amendment #1" or "Contract Amendment #1 - Budget v2" | Review existing AMRs in Award Workspace for consistency of title format | 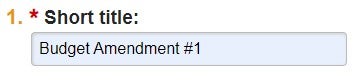 |
| Date Requested | Date of Submission/Request | 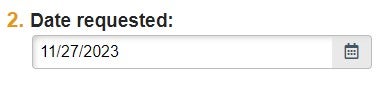 |
||
| Full Description | Include all information needed to complete the subsequent Mod request. | 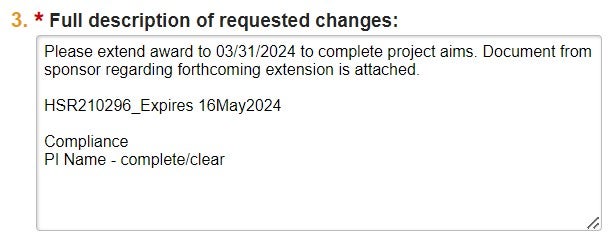 |
||
| 1. Describe full request details, including specific Terms & Conditions being amended. | "Budget Amendment #1 to reflect changes from Protocol version 4." | Include reference to Legacy rUVA record, if applicable. | ||
| 2. Updated IRB Approval & Updated SBCA, if applicable. | "IRB #12345 attached" or "No updated SBCA needed" | SBCA = Study Billing Coverage Analysis | ||
| 3. If necessary, contact info for Sponsor communication. | This is required for all CTA or other bilateral agreements/amendments or if we are submitting a request to the Sponsor for the action. | |||
| 4. Compliance of all S/K Personnel | "Compliance: Person 1 - Complete/Clear Person 2 - Complete [SFI flag] Person 3 - Complete [EAD flag]" | Review personnel compliance via QlikSense "Research Conflicts of Interest" report. Please see instructions for accessing this report here. | ||
| Supporting Documents | 1. Draft contract amendment and supporting documents | File naming convention: "001 - DRAFT - CTA Amendment" | Other possible contracting documents: draft Budget, updated Protocol, original FE CTA [for reference], updated ICF, etc | 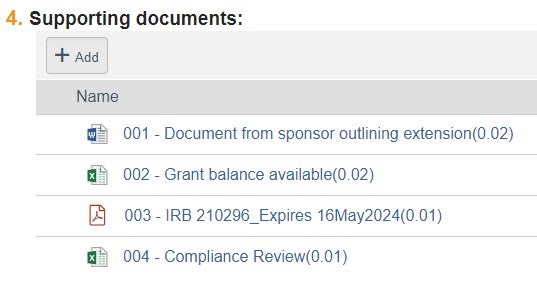 |
| 2. IRB approval | File naming convention: "002 - IRB #12345_Expires 01Jan2023" | |||
| 3. Updated SBCA | File naming convention: "003 - SBCA_Date" | |||
| 4. Compliance printout from QlikSense | File naming convention: "004 - Compliance Review" | |||
| Submitter | Assign Submitter as your SOMOGC administrator | DO NOT click "Submit to Specialist" | 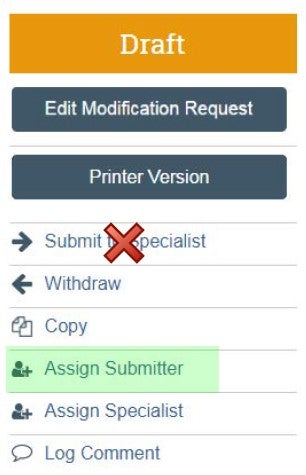 |
|
| Request Type [dropdown] | Other Modification | 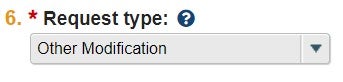 |
||
| General Tips/Notes | Send AMR # and/or link to somogc@uvahealth.org to get the action logged into the SOMOGC work queue. | Without this email, we will have no idea that your AMR is ready for review | ||
| PI and Department approvals should be captured via "Log Comment" activity. | If necessary, approvals can be provided outside of the system and uploaded as supporting documentation. See template here. | 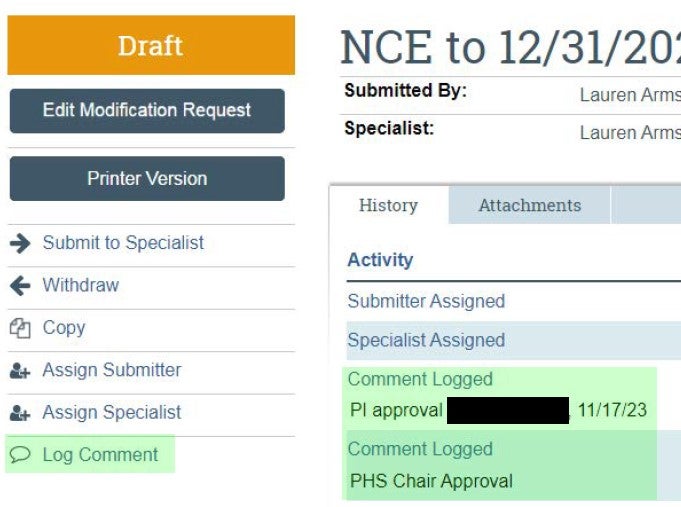 |
||
| For all AMR types, please include relevant email communication thread to provide context for the action being requested. | ||||
| Include all relevant attachments within SmartForm request page, not through "Add Comment" or on History tab. | ||||
| Don't use the WITHDRAW activity unless you really want to discard/trash the action. It cannot be reinstated once withdrawn. |
