Christopher R. McCartney, MD
 PRIMARY APPOINTMENT:
PRIMARY APPOINTMENT:
Professor of Medicine, Endocrinology, and Metabolism
TRAINING:
- Undergraduate: B.S. (Biology), Mississippi College, Clinton, MS
- Graduate: M.D., University of Mississippi School of Medicine, Jackson, MS
- Internal Medicine Residency, University of Mississippi Medical Center, Jackson, MS
- Endocrinology Fellowship, University of Virginia Health System, Charlottesville, VA
- Graduate: M.S. (Health Evaluation Sciences/Clinical Research), University of Virginia Health System
CONTACT:
UVA Division of Endocrinology & Metabolism
PO Box 801406
Charlottesville, VA 22908
Telephone: 434.924.1825
Fax: 434.924.9616
Email: cm2hq@virginia.edu
RESEARCH INTEREST:
Reproductive neuroendocrinology and polycystic ovary syndrome.
RESEARCH DESCRIPTION:
The long-term goal of our clinical (human subject) research is to understand the cause(s) of abnormal gonadotropin-releasing hormone (GnRH) pulses and abnormal gonadotropin secretion in adolescents and adults with polycystic ovary syndrome (PCOS). Our group’s specific goals include elucidation of progesterone’s role in directing GnRH/LH secretion and 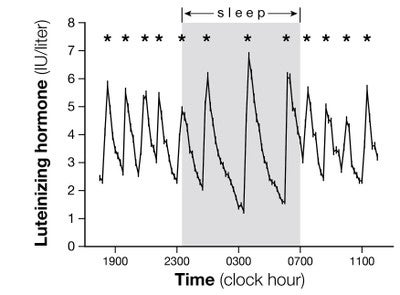 mechanisms controlling wake vs. sleep-associated GnRH/LH pulse frequency in peripubertal girls and women, both in the presence and absence of hyperandrogenemia. Using protocols of frequent hormone sampling and formal sleep analysis, we are testing a working model that involves the primacy of sex steroid negative feedback in the control of GnRH/LH pulse frequency when awake, but the relative inability of sex steroids to influence GnRH/LH pulse frequency during sleep. In addition, we are pursuing studies to assess potential defects in sex steroid-mediated gonadotropin surge generation (positive feedback) in adolescents/women with hyperandrogenemia/PCOS. Moreover, we continue to pursue studies designed to elucidate mechanisms underlying obesity-associated hyperandrogenemia in peripubertal girls.
mechanisms controlling wake vs. sleep-associated GnRH/LH pulse frequency in peripubertal girls and women, both in the presence and absence of hyperandrogenemia. Using protocols of frequent hormone sampling and formal sleep analysis, we are testing a working model that involves the primacy of sex steroid negative feedback in the control of GnRH/LH pulse frequency when awake, but the relative inability of sex steroids to influence GnRH/LH pulse frequency during sleep. In addition, we are pursuing studies to assess potential defects in sex steroid-mediated gonadotropin surge generation (positive feedback) in adolescents/women with hyperandrogenemia/PCOS. Moreover, we continue to pursue studies designed to elucidate mechanisms underlying obesity-associated hyperandrogenemia in peripubertal girls.
Our work is funded by NIH R01 HD102060 (Eunice Kennedy Shriver NICHD, “Role of Androgens in the Neuroendocrine Dysfunction of Nascent PCOS,” September 2019–June 2024).
Recent research fellows have included Su Hee Kim, MD (previously supported by NIH F32 HD088047 [2017-2018] and now supported as a faculty member by NIH K23 HD098319 [2019-2024] and Jessica Lundgren, MD (supported by NIH F32 HD091951 [2017-2019]). Although our trainees contribute to all ongoing studies, prospective new fellows could focus on one of the following areas: (1) the impact of sleep and hyperandrogenemia on sex steroid (progesterone) negative feedback regulation of GnRH pulse frequency; (2) the impact of hyperandrogenemia on estradiol- and progesterone-mediated gonadotropin surge generation (positive feedback); and (3) mechanisms underlying obesity-associated hyperandrogenemia.
