Epigenetics
About
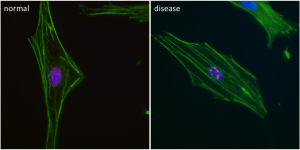 Research in Epigenetics at UVA focuses on the study of heritable changes in phenotype and gene function that are not caused by direct alterations in DNA sequence. DNA in eukaryotic cells is packaged into chromatin by histone proteins. DNA-driven cellular processes such as gene expression require alteration of chromatin structure to access this packaged DNA. Epigenetic changes, including chromatin remodeling, histone exchange, or chemical modification of histone proteins, affect access to chromatin DNA. Modifications to the chromatin through processes such as DNA methylation and histone acetylation are being increasingly appreciated as key determinants of cellular phenotype. Epigenetic chromatin modification is also influenced by environmental factors and can be stably transmitted across generations.
Research in Epigenetics at UVA focuses on the study of heritable changes in phenotype and gene function that are not caused by direct alterations in DNA sequence. DNA in eukaryotic cells is packaged into chromatin by histone proteins. DNA-driven cellular processes such as gene expression require alteration of chromatin structure to access this packaged DNA. Epigenetic changes, including chromatin remodeling, histone exchange, or chemical modification of histone proteins, affect access to chromatin DNA. Modifications to the chromatin through processes such as DNA methylation and histone acetylation are being increasingly appreciated as key determinants of cellular phenotype. Epigenetic chromatin modification is also influenced by environmental factors and can be stably transmitted across generations.
Improved understanding of epigenetic processes is producing exciting advances in cancer detection and treatment. For example, epigenetic profiles can improve cancer detection, and new anti-cancer drugs target the chromatin modifying machinery.
The UVA epigenetics research groups utilize model organisms, mammalian cells and human-derived samples in their studies. A variety of experimental approaches, including biochemistry, genetics, genome-wide analysis and bioinformatics, and behavioral studies are being applied to study epigenetic mechanisms and changes associated with human disease.
Faculty
Bekiranov, Stefan
Computational Biology; Bioinformatics; Precision Medicine; Machine Learning/AI; Quantum Computing
Bushweller, John H.
Drug Development Targeting Transcription Drivers in Cancer; Structure/Function Studies of Transcription Factor Drivers in Cancer
Cliffe, Anna
Herpes Simplex Virus Infection of Neurons
Connelly, Jessica J.
Epigenetic mechanisms involved in complex human disease
Gan, Lu
Chromosome organization; Nucleosome structure; Histone marks; Structural cell biology
Garrett-Bakelman, Francine E.
Acute Myeloid Leukemia: molecular and cellular biology events which mediate aberrant epigenetic and transcriptional mechanisms during disease establishment and progression
Goldfarb, Adam N.
Hematopathology and Understanding the Molecular Basis of Hematopoiesis and Leukemogenesis
Guo, Lian-Wang
Vascular wall remodeling, intimal hyperplasia, stenosis, aneurysm, retinal degeneration.
Harris, Thurl E
Molecular mechanisms controlling insulin signaling and fat synthesis.
Liu, Wenpeng
Genetic and Epigenetic stability in human cancer
Loughran, Jr., Thomas P
Hematologic malignancies; bone marrow disorders; leukemia; large granular lymphocyte (LGL)
Lynch, Wendy J.
Behavioral Pharmacology, Sex Differences, Animal Models of Addiction
Mayo, Marty W.
Transcriptional Regulation by NFKB
Miller, Clint L.
Genetic variation, Complex diseases, Coronary artery disease, Genomics, Epigenomics, Regulatory mechanisms, Vascular biology, Pharmacology and Physiology
Mohi, Golam
Cell signaling, stem cell biology, molecular and epigenetic mechanisms of blood and breast cancers
Owens, Gary K.
Identification of Factors and Mechanisms that Regulate the Stability of Late Stage Atherosclerotic Lesions and the Probability of Thromboembolic Events Including a Heart Attack or Stroke
Park, Kwon
Mechanisms of tissue/organ homeostasis and tumorigeensis
Pemberton, Lucy F.
Director of Graduate Studies for Microbiology, Immunology and Cancer Biology
Puglia, Meghan
Developmental Cognitive Neuroscience
Rekosh, David M.
Human Immunodeficiency Virus Gene Expression; Human Endogenous Viruses; SARS-CoV-2 Protein Trafficking; Post-transcriptional Gene Regulation
Rich, Stephen S.
Genetic basis of common human disease, including type 1 diabetes, diabetic complications, ischemic stroke, atherosclerosis
Sheffield, Nathan
computational biology & bioinformatics; high performance computing; epigenomics & chromatin; pediatric cancer; computational regulatory genomics; machine learning
Smith, Jeffrey S.
Transcriptional Silencing and Aging in Yeast
Stukenberg, P. Todd
Mechanisms of chromosome segregation in Mitosis and generation of Chromosomal Instability in tumors
Tao, Jianguo
Pathology
Trinh, Bon Q
Understanding Protein and RNA regulations of gene expression via chromatin structure in myeloid cell development and diseases
Wang, Yuh-Hwa
Genome instability in cancer and repeat expansion diseases
Weaver, Tyler M.
Mechanisms of DNA repair in chromatin
Wotton, David
Regulation of Gene Expression, Development and Tumor Progression by TGF beta Signaling
Xu, Kexin
Researching fundamental roles of the epigenome in control of the context-specific transcriptional programs.
Zang, Chongzhi
Bioinformatics methodology development; Epigenetics and chromatin biology; Transcriptional regulation; Cancer genomics and epigenomics; Statistical methods for biomedical data integration; Advanced machine learning; Theoretical and computational biophysics
Zhou, Anny Xiaobo
Genetics, Functional Genomics, Epigenetics, Lung repair/regeneration, Metabolic regulation, Alveolar stem cells, Chromatin interaction, Innate immune response, Live-cell imaging
